 Mother-to-child transmission of HIV through breastfeeding is a significant source of new infections in the developing world - accounting for as many as half of the 350,000 new pediatric infections each year world wide.
Mother-to-child transmission of HIV through breastfeeding is a significant source of new infections in the developing world - accounting for as many as half of the 350,000 new pediatric infections each year world wide.
But in developing countries not breastfeeding is even more dangerous to infant health and survival, dramatically increasing the risk of death from infectious diseases. So for some time the recommendation has been, in various forms, that HIV positive mothers in developing countries formula feed only if it is “affordable, feasible, safe, and sustainable.” Otherwise, exclusive breastfeeding has been recommended.
The relatively recent finding that anti-retroviral drugs (ARVs) administered during pregnancy and lactation dramatically reduce transmission risk led to the current WHO recommendation for HIV positive mothers in developing countries: “women who breastfeed and receive ARVs (or whose infants are receiving ARVs) should exclusively breastfeed their infants for 6 months and continue breastfeeding until 12 months of age and only then consider stopping. In developed countries, the recommendation has been to exclusively formula feed (though see the final study listed here on this topic).
But there remain many mysteries about HIV and breastfeeding: Why do most infants not contract the virus through breastfeeding when some do? What is it in milk which prevents infection? Why does mixed feeding carry the highest risk? How is the range of viral load related to transmission? Most importantly, what is the most effective way of preventing transmission while preserving breastfeeding?
2012 brought quite a bit of new research to bear on these and other questions. I am far from an expert on this topic, but given the large volume of research published on this topic this year, I thought it would be a good idea to offer some highlights:
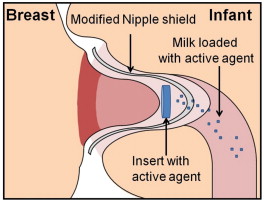
Could a nipple shield containing a chemical which inactivates HIV prevent transmission through breastfeeding? This study found that the use of a nipple shield with a chemical insert (see illustration above): “Greater than 99% reduction of cell-associated HIV infectivity was achieved in the first 10 ml of milk. This proof of concept study demonstrates efficient drug delivery to breastfeeding infants is achievable using the nipple shield delivery system.”
Oligosaccharides may be partially responsible for inhibiting transmission (Zambia): “The inefficiency of HIV breast-milk transmission may be caused by the presence of immunologically active factors, including human milk oligosaccharide (HMOs). HIV-infected women with total HMOs above the median (1.87 g/L) were less likely to transmit via breastfeeding.”
Could flash-heating of breastmilk (which kills the HIV virus) be used in resource-poor settings? (Tanzania): “Flash heating is a simple technology that many HIV-positive women can successfully use after exclusive breastfeeding to continue to provide the benefits of breastmilk while avoiding maternal-to-child transmission associated with nonexclusive breastfeeding. Based on these feasibility data, a clinical trial of the effects of flash heating breastmilk on infant health outcomes is warranted.” (I actually donated breastmilk for an early version of this study!)
B cells in milk can generate HIV-neutralizing antibodies. “Our work helped establish that these B cells in breast milk can produce HIV-neutralizing antibodies, so enhancing the response or getting more mucosal B-cells to produce those helpful antibodies would be useful, and this is a possible route to explore for HIV-1 vaccine development.”
Does mixed feeding lead to subclinical mastitis and higher viral load in breastmilk, accounting for the higher risk of infection with mixed feeding? (Botswana): “We report no observed relationship between mixed breastfeeding and measured breast milk viral RNA load. Our findings suggest that the increased transmission risk associated with higher breast milk HIV-1 RNA during mixed breastfeeding is unlikely.”
But inflammation might be important depending upon the type of infection: “Enhancement of cell-associated HIV-1 infection by breast milk may be associated with inflammatory conditions in the mother and may contribute to infant infection during breastfeeding.”
Children infected perinatally are at higher risk of dying than those infected through breastmilk (multiple studies): “One year after acquisition of HIV infection, an estimated 26% postnatally and 52% perinatally infected children would have died; and 4% uninfected children by age 1 year.”
In an animal study, breastmilk could be induced to become more HIV-inhibiting. “The neutralizing activity detected in milk following systemic administration of a broadly- neutralizing IgG antibody supports the induction of strong systemic anti-HIV IgG responses to generate HIV inhibitory antibodies in breast milk.” Or perhaps through genetic engineering?
A surprisingly high number of mothers become infected with HIV in between their first prenatal visit and weaning; testing later in pregnancy and during breastfeeding could make a big difference (South Africa): “The proportion of mother-to-child transmission (MTCT) from mothers who seroconverted after their first antenatal visit was 26%. If maternal HIV incidence during late pregnancy and breastfeeding were reduced by 50% after 2010, and HIV screening were repeated in late pregnancy and at 6-week immunization visits after 2010, the average annual number of MTCT cases over the 2010–2015 period would reduce by 28%, from 39,000 to 28,000 per annum.”
When mothers take ARVs it dramatically reduces the infection risk in their babies. What about if the babies take it, too (or instead)? (South Africa, Tanzania, Uganda, Zimbabwe): “Once-daily nevirapine prophylaxis [given to infant] reduces mother-to-child HIV-1 transmission via breast milk up to age 6 months, especially if the mother is not taking antiretroviral therapy.” And it appears to be safe for the infant, too.
Still more evidence of the importance of exclusive breastfeeding when alternatives are not affordable, feasible, safe, and sustainable (Tanzania): “Longer exclusive breastfeeding (EBF) by HIV-positive mothers was associated with reduced mortality in the first 6 mo of life without increased HIV infection. A 1-mo increase in EBF was associated with a 49% reduction in early infant mortality in the first 6 mo of life.” And another (South Africa): “Breastfeeding infants had a significantly lower risk of diarrhoea and hospitalisation at 3 months. Breastfeeding was significantly associated with better development scores and growth parameters.”
Is if safe for HIV positive mothers in developed countries to breastfeed under certain circumstances? (U.K.): In 2010 the British HIV Association (BHIVA) published a position statement (not an official practice guideline) which states that in the U.K., HIV positive mothers should formula feed from birth, but: “In the very rare instances where a mother in the UK who is on effective HAART with a repeatedly undetectable viral load chooses to breast feed, BHIVA/CHIVA …do not regard this as grounds for automatic referral to child protection teams. Maternal HAART should be carefully monitored and continued until one week after all breastfeeding has ceased. Breastfeeding, except during the weaning period, should be exclusive and all breastfeeding, including the weaning period, should have been completed by the end of 6 months.”
A study published this year looked at the viral loads of 41 U.K. women on ARVs, and found: “Of those who were meant to be undetectable on ART, 6/28 (21.4%) had a viral load > 100 copies/ml. 12(92.3%) of those who took ART for PMTCT were detectable at post-delivery viral load. Our clinic review suggests that if breast feeding is to be supported in certain circumstances: i) increased frequency of monitoring will be necessary for those on ART; ii) those on ART for PMTCT only would need to continue ART in the post natal period with such monitoring.”